Research
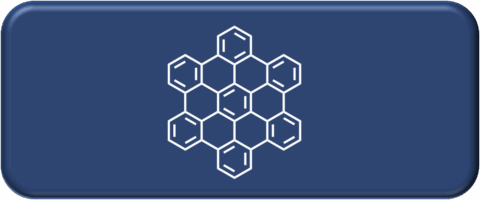
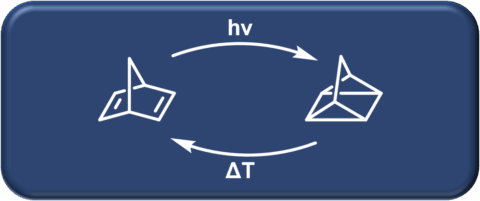
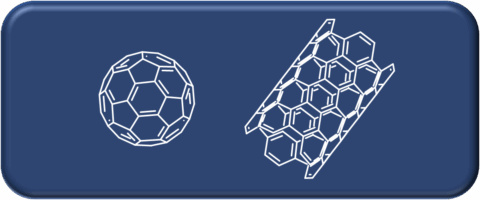
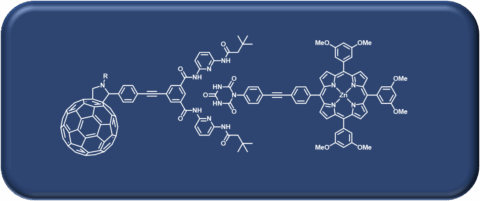
The Hirsch laboratory pioneered the chemistry of the synthesis and functionalization of carbon allotropes, ranging from fullerenes to carbon nanotubes to graphene. Further research interests include synthesizing various other carbon-based materials for solar energy conversion and storage, catalysis, and supramolecular chemistry.